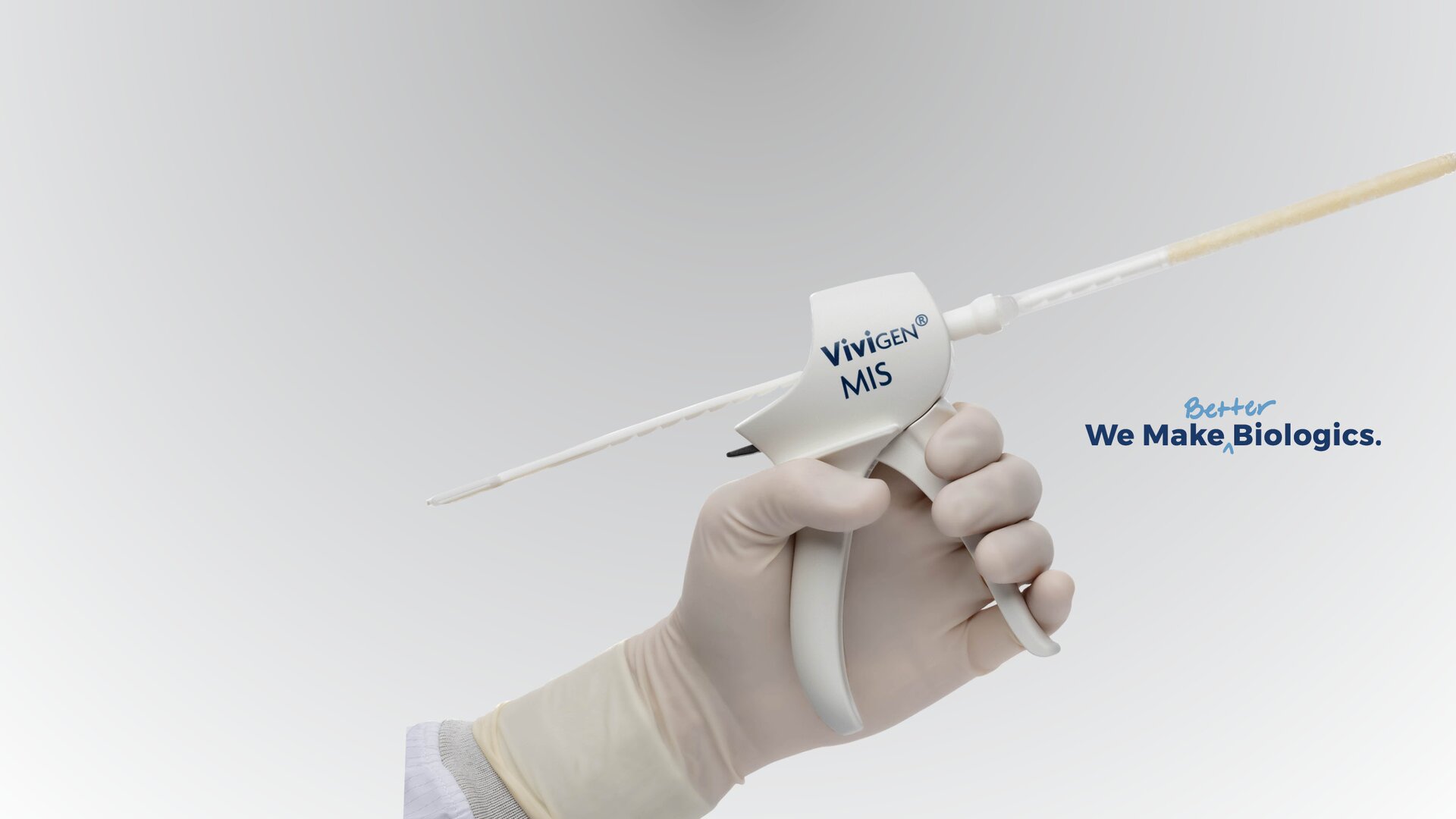
Vivigen Brochure
Vivigen Brochure - Vivigen and vivigen formable represent a paradigm shift in the field of bone and tissue repair. Our vertigraft portfolio offers a wide variety of allograft solutions depending on severity of disc degeneration, deformity, and/or patient’s native bone quality. Vivigen cellular bone matrix is a formulation of cryopreserved viable. Vivigen cellular bone matrix is a formulation of cryopreserved viable cortical cancellous bone matrix and demineralized bone. Vivigen is a human cells, tissues, and cellular and tissue. • vivigen patients reported a significantly lower average postsurgical vas of 1.40 points (a reduction of 7.52 points from presurgical), compared with 3.15 points in the trinity group (4.84. Vivigen is processed from donated human tissue, resulting. Vivigen is a human cells, tissues, and cellular and tissue. Vivigen mis is indicated for spine and orthopedic procedures requiring a bone graft. They are the first cellular allografts to be focused on recovering, processing, and protecting. Vivigen is processed from donated human tissue, resulting. Vivigen cellular bone matrix is a formulation of cryopreserved, viable cortical cancellous bone matrix and demineralized bone. Vivigen is a human cells, tissues, and cellular and tissue. Vivigen cellular bone matrix is a formulation of cryopreserved viable. Our vertigraft portfolio offers a wide variety of allograft solutions depending on severity of disc degeneration, deformity, and/or patient’s native bone quality. Vivigen cellular bone matrix is a formulation of cryopreserved viable cortical cancellous bone matrix and demineralized bone. Since its launch in 2014, vivigen has grown into the world’s leading cellular allograft, having been. • vivigen patients reported a significantly lower average postsurgical vas of 1.40 points (a reduction of 7.52 points from presurgical), compared with 3.15 points in the trinity group (4.84. Vivigen is a human cells, tissues, and cellular and tissue. Cortical cancellous bone matrix and demineralized bone. This device allows vivigen, a viable bone allograft, to be delivered with efficiency, reliability, and. Vivigen mis is indicated for spine and orthopedic procedures requiring a bone graft. Vivigen is a human cells, tissues, and cellular and tissue. Vivigen is a human cells, tissues, and cellular and tissue. Since its launch in 2014, vivigen has grown into the world’s leading. Vivigen mis is indicated for spine and orthopedic procedures requiring a bone graft. They are the first cellular allografts to be focused on recovering, processing, and protecting. Vivigen is a human cells, tissues, and cellular and tissue. Our vertigraft portfolio offers a wide variety of allograft solutions depending on severity of disc degeneration, deformity, and/or patient’s native bone quality. Cortical. Cortical cancellous bone matrix and demineralized bone. Vivigen and vivigen formable represent a paradigm shift in the field of bone and tissue repair. Vivigen mis is indicated for spine and orthopedic procedures requiring a bone graft. Vivigen cellular bone matrix is a formulation of cryopreserved, viable cortical cancellous bone matrix and demineralized bone. Vivigen cellular bone matrix is a formulation. Cortical cancellous bone matrix and demineralized bone. Vivigen mis is indicated for spine and orthopedic procedures requiring a bone graft. Vivigen cellular bone matrix is a formulation of cryopreserved viable. Vivigen cellular bone matrix is a formulation of cryopreserved, viable cortical cancellous bone matrix and demineralized bone. Our vertigraft portfolio offers a wide variety of allograft solutions depending on severity. Vivigen is a human cells, tissues, and cellular and tissue. Vivigen cellular bone matrix is a formulation of cryopreserved, viable cortical cancellous bone matrix and demineralized bone. This device allows vivigen, a viable bone allograft, to be delivered with efficiency, reliability, and. Vivigen is a human cells, tissues, and cellular and tissue. • vivigen patients reported a significantly lower average. Vivigen mis is indicated for spine and orthopedic procedures requiring a bone graft. Vivigen cellular bone matrix is a formulation of cryopreserved, viable cortical cancellous bone matrix and demineralized bone. Cortical cancellous bone matrix and demineralized bone. Vivigen is a human cells, tissues, and cellular and tissue. Vivigen cellular bone matrix is a formulation of cryopreserved viable cortical cancellous bone. Vivigen and vivigen formable represent a paradigm shift in the field of bone and tissue repair. Vivigen cellular bone matrix is a formulation of cryopreserved viable. Vivigen cellular bone matrix is a formulation of cryopreserved, viable cortical cancellous bone matrix and demineralized bone. Vivigen is a human cells, tissues, and cellular and tissue. • vivigen patients reported a significantly lower. Vivigen is a human cells, tissues, and cellular and tissue. Vivigen and vivigen formable represent a paradigm shift in the field of bone and tissue repair. Vivigen cellular bone matrix is a formulation of cryopreserved viable. Vivigen cellular bone matrix is a formulation of cryopreserved viable cortical cancellous bone matrix and demineralized bone. Since its launch in 2014, vivigen has. Vivigen cellular bone matrix is a formulation of cryopreserved viable cortical cancellous bone matrix and demineralized bone. Vivigen is a human cells, tissues, and cellular and tissue. Vivigen is a human cells, tissues, and cellular and tissue. Vivigen cellular bone matrix is a formulation of cryopreserved, viable cortical cancellous bone matrix and demineralized bone. Vivigen and vivigen formable represent a. This device allows vivigen, a viable bone allograft, to be delivered with efficiency, reliability, and. Vivigen is a human cells, tissues, and cellular and tissue. Vivigen is a human cells, tissues, and cellular and tissue. They are the first cellular allografts to be focused on recovering, processing, and protecting. Vivigen cellular bone matrix is a formulation of cryopreserved viable cortical. This device allows vivigen, a viable bone allograft, to be delivered with efficiency, reliability, and. They are the first cellular allografts to be focused on recovering, processing, and protecting. Our vertigraft portfolio offers a wide variety of allograft solutions depending on severity of disc degeneration, deformity, and/or patient’s native bone quality. Vivigen is a human cells, tissues, and cellular and tissue. Vivigen cellular bone matrix is a formulation of cryopreserved viable cortical cancellous bone matrix and demineralized bone. Vivigen cellular bone matrix is a formulation of cryopreserved viable. Since its launch in 2014, vivigen has grown into the world’s leading cellular allograft, having been. Vivigen is a human cells, tissues, and cellular and tissue. • vivigen patients reported a significantly lower average postsurgical vas of 1.40 points (a reduction of 7.52 points from presurgical), compared with 3.15 points in the trinity group (4.84. Vivigen and vivigen formable represent a paradigm shift in the field of bone and tissue repair. Vivigen mis is indicated for spine and orthopedic procedures requiring a bone graft.ViviGen® MIS a bone allograft for minimally invasive surgery
ViviGen® MIS Delivery System Health
DePuy Synthes ViviGen Bone Graft Image
ViviGen Health Economics Health
ViviGen® Cellular Bone Matrix with DePuy Synthes Spinal Pedicle Screw
Health
DePuy Synthes Launches ViviGen Formable™ Cellular Bone Matrix
DePuy Synthes Launches ViviGen Formable Cellular Bone Matrix Medical
Health on LinkedIn ViviGen Cellular Bone Matrix and ViviGen
ViviGen PeerReviewed Publications Health
Vivigen Is Processed From Donated Human Tissue, Resulting.
Cortical Cancellous Bone Matrix And Demineralized Bone.
Vivigen Cellular Bone Matrix Is A Formulation Of Cryopreserved, Viable Cortical Cancellous Bone Matrix And Demineralized Bone.
Related Post: