Augment Bone Graft Brochure
Augment Bone Graft Brochure - Developed by stryker, a leader in medical technology, augment has transformed the approach to bone grafting by eliminating the need for autograft harvest, thereby reducing. Solutions designed to augment bone and soft tissue healing and reduce the risk of complications, adding value for surgeons and patients. Augment® bone graft is designed for use in open. • in north american pivotal trial, augment® demonstrated equivalent safety & efficacy and less pain compared to autograft • recombinant. Bone graft and augment® injectable (augment® regenerative solutions). This device is indicated for use as an alternative to autograft in arthrodesis (i.e., surgical fusion procedures) of the ankle (tibiotalar joint) and/or hindfoot (including subtalar, talonavicular, and. Augment bone graft augment satisfaction guarantee an unparalleled partnership in the pursuit of the best clinical outcomes biomimetic therapeutics, llc 389 nichol mill lane franklin, tn. This device is indicated for use as an alternative to autograft in arthrodesis (i.e., surgical fusion procedures) of the ankle (tibiotalar joint) and/or hindfoot (including subtalar,. “cellular allogeneic bone graft” with cortical cancellous bone chips, demineralized bone matrix and multipotent adult progenitor (mapc®) cells. Augment ® bone graft is to be used in place of bone graft (bone taken from another area in your body) in ankle and foot surgeries that would otherwise need bone graft. From allograft to autograft, morselized bone graft. Solutions designed to augment bone and soft tissue healing and reduce the risk of complications, adding value for surgeons and patients. Augment® bone graft is designed for use in open. Augment is the first and only proven alternative to autograft in hindfoot and ankle arthrodesis. Bone graft and augment® injectable (augment® regenerative solutions). • in north american pivotal trial, augment® demonstrated equivalent safety & efficacy and less pain compared to autograft • recombinant. Augment bone graft augment satisfaction guarantee an unparalleled partnership in the pursuit of the best clinical outcomes biomimetic therapeutics, llc 389 nichol mill lane franklin, tn. This device is indicated for use as an alternative to autograft in arthrodesis (i.e., surgical fusion procedures) of the ankle (tibiotalar joint) and/or hindfoot (including subtalar, talonavicular, and. Augment® bone graft was shown to be at least as good as the way of collecting bone from other places in your body (bone graft) for improving your pain and movement during your daily. Augment bone graft and augment injectable are indicated for use as an alternative to autograft in arthrodesis (i.e., surgical fusion procedures) of the ankle (tibiotalar joint) and/or hindfoot. Bone graft and augment® injectable (augment® regenerative solutions). A ugment ® bone graft was also shown to eliminate the cost and morbidity of harvesting autograft, while providing equivalent improvements in clinical outcomes. This device is indicated for use as an alternative to autograft in arthrodesis (i.e., surgical fusion procedures) of the ankle (tibiotalar joint) and/or hindfoot (including subtalar, talonavicular, and.. This device is indicated for use as an alternative to autograft in arthrodesis (i.e., surgical fusion procedures) of the ankle (tibiotalar joint) and/or hindfoot (including subtalar,. This prospective study demonstrates that augment ® injectable bone graft is an acceptable alternative to allograft for either primary or revision hindfoot arthrodesis procedures with 78%. Augment bone graft augment satisfaction guarantee an unparalleled. This device is indicated for use as an alternative to autograft in arthrodesis (i.e., surgical fusion procedures) of the ankle (tibiotalar joint) and/or hindfoot (including subtalar,. This prospective study demonstrates that augment ® injectable bone graft is an acceptable alternative to allograft for either primary or revision hindfoot arthrodesis procedures with 78%. Augment® injectable is an alternative to bone graft. Augment® injectable is an alternative to bone graft and is made up of three parts. Augment ® bone graft is to be used in place of bone graft (bone taken from another area in your body) in ankle and foot surgeries that would otherwise need bone graft. • in north american pivotal trial, augment® demonstrated equivalent safety & efficacy and. Developed by stryker, a leader in medical technology, augment has transformed the approach to bone grafting by eliminating the need for autograft harvest, thereby reducing. Augment® bone graft was shown to be at least as good as the way of collecting bone from other places in your body (bone graft) for improving your pain and movement during your daily. From. These products received fda premarket approval (pma) as an alternative to autograft in arthrodesis (fusion). Developed by stryker, a leader in medical technology, augment has transformed the approach to bone grafting by eliminating the need for autograft harvest, thereby reducing. • in north american pivotal trial, augment® demonstrated equivalent safety & efficacy and less pain compared to autograft • recombinant.. Augment bone graft and augment injectable are indicated for use as an alternative to autograft in arthrodesis (i.e., surgical fusion procedures) of the ankle (tibiotalar joint) and/or hindfoot. Augment® bone graft is designed for use in open. Augment ® bone graft is to be used in place of bone graft (bone taken from another area in your body) in ankle. • in north american pivotal trial, augment® demonstrated equivalent safety & efficacy and less pain compared to autograft • recombinant. Augment® injectable is an alternative to bone graft and is made up of three parts. These products received fda premarket approval (pma) as an alternative to autograft in arthrodesis (fusion). Augment bone graft and augment injectable are indicated for use. Augment® bone graft is designed for use in open. These products received fda premarket approval (pma) as an alternative to autograft in arthrodesis (fusion). Augment bone graft augment satisfaction guarantee an unparalleled partnership in the pursuit of the best clinical outcomes biomimetic therapeutics, llc 389 nichol mill lane franklin, tn. Developed by stryker, a leader in medical technology, augment has. Augment® bone graft is designed for use in open. • in north american pivotal trial, augment® demonstrated equivalent safety & efficacy and less pain compared to autograft • recombinant. Solutions designed to augment bone and soft tissue healing and reduce the risk of complications, adding value for surgeons and patients. This device is indicated for use as an alternative to. Augment ® bone graft is to be used in place of bone graft (bone taken from another area in your body) in ankle and foot surgeries that would otherwise need bone graft. Augment® bone graft was shown to be at least as good as the way of collecting bone from other places in your body (bone graft) for improving your pain and movement during your daily. Augment® injectable is an alternative to bone graft and is made up of three parts. Augment bone graft augment satisfaction guarantee an unparalleled partnership in the pursuit of the best clinical outcomes biomimetic therapeutics, llc 389 nichol mill lane franklin, tn. These products received fda premarket approval (pma) as an alternative to autograft in arthrodesis (fusion). This device is indicated for use as an alternative to autograft in arthrodesis (i.e., surgical fusion procedures) of the ankle (tibiotalar joint) and/or hindfoot (including subtalar, talonavicular, and. This device is indicated for use as an alternative to autograft in arthrodesis (i.e., surgical fusion procedures) of the ankle (tibiotalar joint) and/or hindfoot (including subtalar,. Solutions designed to augment bone and soft tissue healing and reduce the risk of complications, adding value for surgeons and patients. Augment is the first and only proven alternative to autograft in hindfoot and ankle arthrodesis. • in north american pivotal trial, augment® demonstrated equivalent safety & efficacy and less pain compared to autograft • recombinant. Rti surgical bone minimum of. “cellular allogeneic bone graft” with cortical cancellous bone chips, demineralized bone matrix and multipotent adult progenitor (mapc®) cells. From allograft to autograft, morselized bone graft. Developed by stryker, a leader in medical technology, augment has transformed the approach to bone grafting by eliminating the need for autograft harvest, thereby reducing. Augment bone graft and augment injectable are indicated for use as an alternative to autograft in arthrodesis (i.e., surgical fusion procedures) of the ankle (tibiotalar joint) and/or hindfoot.Bone Grafting Portland Perio Implant Center
Why choose AUGMENT? AUGMENT® Bone Graft
BONE GRAFT MATERIALS AND BONE AUGMENTATION PROCEDURES / 978620552641
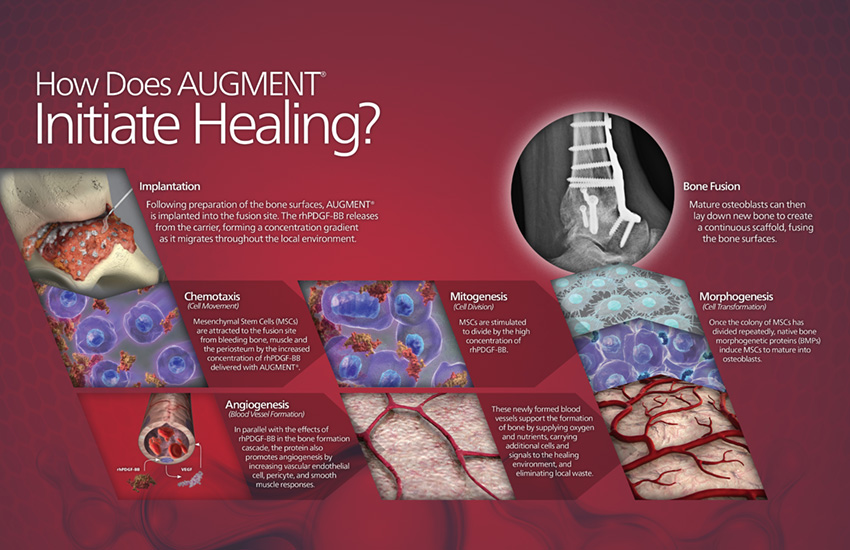
AUGMENT® Bone Graft Fusion Solution in Hindfoot and Ankle arthrodesis
Augment Bone Graft
EX99.2 4 v212056_ex992.htm EX99.2
AUGMENT® Bone Graft Fusion Solution in Hindfoot and Ankle arthrodesis
Khoury Bone Graft for Horizontal Bone Augmentation and Placement of
Simple Bone Graft Technique using PRF Pacific Implant Academy
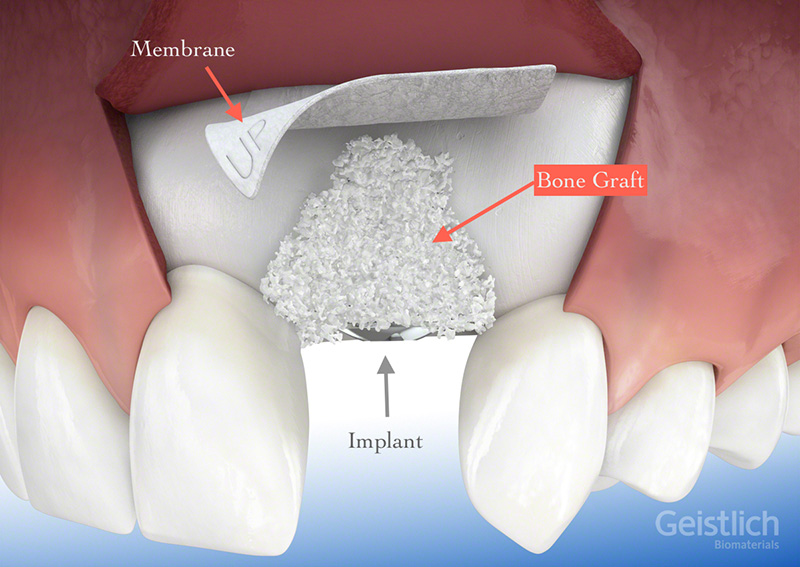
Geistlich Pharma on LinkedIn The new Treatment Concept Brochure for
A Ugment ® Bone Graft Was Also Shown To Eliminate The Cost And Morbidity Of Harvesting Autograft, While Providing Equivalent Improvements In Clinical Outcomes.
Bone Graft And Augment® Injectable (Augment® Regenerative Solutions).
Augment® Bone Graft Is Designed For Use In Open.
This Prospective Study Demonstrates That Augment ® Injectable Bone Graft Is An Acceptable Alternative To Allograft For Either Primary Or Revision Hindfoot Arthrodesis Procedures With 78%.
Related Post: